Fluorescence Simulations
Our downloadable fluorescence simulations create a virtual laboratory with chemicals, glassware and a lifetime spectrometer simulator
Fluorescence Experiment 1: Arrhenius Behaviour
Background
Arrhenius behaviour occurs in many systems in physical chemistry and even in everyday life. The Arrhenius equation relates the rate constant of a chemical reaction to temperature and the activation energy for the process (the energy barrier that must be overcome for reaction to proceed.) It’s why we store food in refrigerators: since rotting food is a chemical reaction we can slow the rate of decay down by storing in a cool environment. The fluorescence emission of certain aromatic molecules when dissolved in solution will also show an Arrhenius type thermal dependence. Our simulation software allows the student to prepare a luminescent solution and observe its fluorescence decay over the temperature range 20 ºC to 60 ºC via a fluorescence lifetime spectrometer. Ultimately, the rate constants for fluorescence and the activation energy for the process can be derived.
Fluorescence Experiment 1, Arrhenius Behaviour
In the first part of this experiment, stock solutions and dilutions will be prepared for fluorescently labelled polymer samples using the virtual flask module. Fluorescence and UV-Vis absorption spectra will also be recorded:
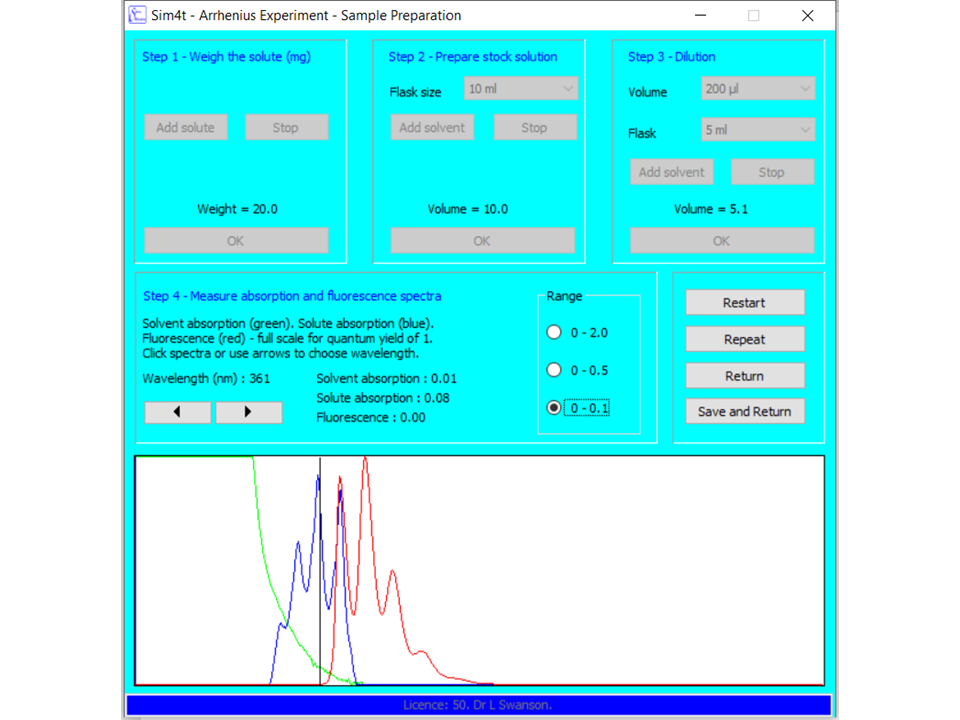
A fluorescence decay will be recorded at room temperature using the single photon counter simulator with temperature control. These decay measurements will be repeated at several different temperatures.

A corresponding instrument profile (prompt) will be recorded following each decay curve measurement:

A fluorescence lifetime will be derived at each temperature using our decay curve analyses software:

The derived lifetimes will be converted into rate constants at each temperature and plotted to derive the Arrhenius parameter and Activation Energy for the process.
Any questions? Please contact our experts who will be happy to help.
We cover a variety of topics using a UV-Vis Absorption spectrometer simulator: Everything from studying the kinetics of the crystal violet/sodium hydroxide reaction to determining the level of aspirin in a blood sample. Take a look at our product page.
